
Emily Balskus (left), Morris Kahn Associate Professor of Chemistry and Chemical Biology, looks over the shoulder of graduate student Vayu Maini Rekdal, working with microbes in an oxygen-free environment.
Photos by Jon Chase/Harvard Staff Photographer
Gut microbes eat our medication
How one species of bacteria consumes the primary treatment for Parkinson’s disease could reveal more about how the microbiome impacts our health
The first time a young Vayu Maini Rekdal manipulated microbes, he made a decent sourdough bread, even if he gave little thought to the crucial chemical reactions involved.
More crucial, he would later learn, is the role microbes play in helping our bodies break down foods so they can absorb the nutrients. Since we cannot digest certain substances — all-important fiber, for example — microbes step up to perform chemistry no human can.
“But this kind of microbial metabolism can also be detrimental,” said Maini Rekdal, a Graduate School of Arts and Sciences Ph.D. student in the lab of Professor Emily Balskus, and first author on their new study published June 14 in Science.
According to Maini Rekdal, gut microbes can chew up medications with often hazardous side effects. “Maybe the drug is not going to reach its target in the body; maybe it’s going to be toxic all of a sudden; maybe it’s going to be less helpful,” he said.
In their study, Balskus, Maini Rekdal, and their collaborators at the University of California, San Francisco, describe one of the first concrete examples of how the microbiome can interfere with a drug’s intended path through the body. Focusing on levodopa (L-dopa), the primary treatment for Parkinson’s disease, they identified which bacteria out of the trillions of species is responsible for degrading the drug, and how to stop it.
Parkinson’s disease globally affects more than 1 percent of those age 60 and above. The neurological disorder attacks nerve cells in the brain that produce dopamine, without which the body can suffer tremors, muscle rigidity, and problems with balance and coordination. The cause of the disease is unknown.
The primary treatment for Parkinson’s symptoms is L-dopa, a drug taken orally that delivers dopamine to the brain. To do so, it must first cross the blood-brain barrier. For most patients, only about 1 to 5 percent of the drug actually reaches the brain.
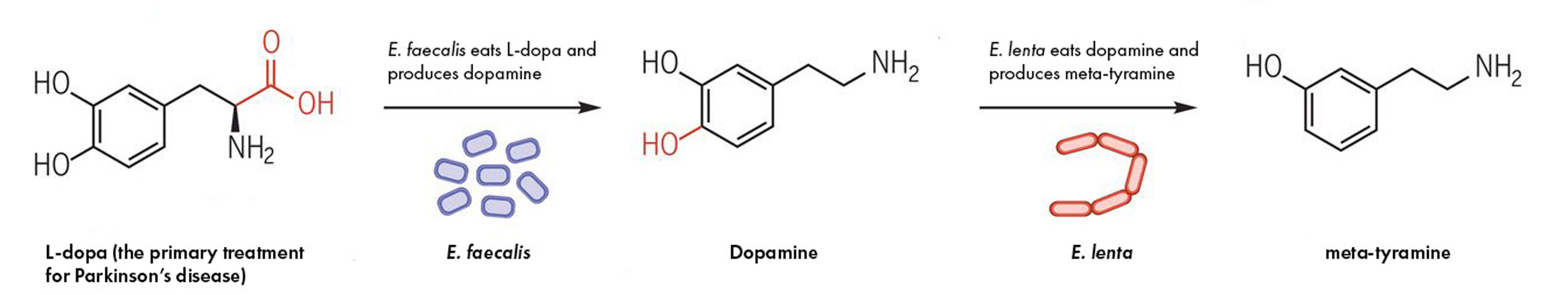
The two species of gut bacteria involved in metabolizing the Parkinson’s drug, L-dopa. First, E. faecalis eats L-dopa and spits out dopamine. Then, E. lenta eats dopamine and spits out meta-tyramine. The first step contributes to drug loss but both could contribute to L-dopa’s sometimes severe side effects.
Source: “Discovery and inhibition of an interspecies gut bacterial pathway for Levodopa metabolism”
This number — and the drug’s efficacy — varies widely from patient to patient. Since the introduction of L-dopa in the late 1960s, researchers have known that the body’s enzymes (tools that perform necessary chemistry) can break down L-dopa in the gut, preventing the drug from reaching the brain. The pharmaceutical industry introduced aneffective supplemental drug, carbidopa, to block unwanted L-dopa metabolism.
“Even so,” Maini Rekdal said, “there’s a lot of metabolism that’s unexplained, and it’s very variable between people.” That sets the stage for another problem: Not only is the drug less effective for some patients, but when L-dopa is transformed into dopamine outside the brain, the compound can cause side effects, including severe gastrointestinal distress and cardiac arrhythmias. If less of the drug reaches the brain, patients are often given more to manage their symptoms, potentially worsening the side effects.
Maini Rekdal suspected microbes might be behind the L-dopa disappearance. Previous research showed that antibiotics improve a patient’s response to L-dopa, but scientists could only speculate that bacteria might be to blame. Still, no one identified which bacterial species might be culpable or how and why they eat the drug.
So the Balskus team launched an investigation. The unusual chemistry — L-dopa to dopamine — was their first clue. Few bacterial enzymes can perform this conversion. But a good number bind to tyrosine — an amino acid similar to L-dopa. And one, from a food microbe often found in milk and pickles (Lactobacillus brevis), can accept both tyrosine and L-dopa.


Vayu Maini Rekdal, shown handling microbes in an oxygen-free environment, works with Emily Balskus in the lab.
Using the Human Microbiome Project as a reference, Maini Rekdal and his team hunted through bacterial DNA to identify which gut microbes had genes to encode a similar enzyme. Several fit their criteria, but only one strain, Enterococcus faecalis (E. faecalis), ate all the L-dopa every time.
With this discovery, the team provided the first strong evidence connecting E. faecalis and the bacteria’s enzyme (PLP-dependent tyrosine decarboxylase, or TyrDC) to L-dopa metabolism. Yet a human enzyme can and does convert L-dopa to dopamine in the gut, the same reaction carbidopa is designed to stop. Then why, the team wondered, does the E. faecalis enzyme escape carbidopa’s reach?
Even though the human and bacterial enzymes perform the exact same chemical reaction, the bacterial one looks just a little different. Maini Rekdal speculated that carbidopa may not be able to penetrate the microbial cells, or the slight structural variance could prevent the drug from interacting with the bacterial enzyme. If true, other host-targeted treatments may be just as ineffective as carbidopa against similar microbial machinations.
But the cause may not matter. Balskus and her team already discovered a molecule capable of inhibiting the bacterial enzyme.
“The molecule turns off this unwanted bacterial metabolism without killing the bacteria; it’s just targeting a nonessential enzyme,” Maini Rekdal said. This and similar compounds could provide a starting place for the development of new drugs to improve L-dopa therapy for Parkinson’s patients.
The team might have stopped there. Instead, they unraveled a second step in the microbial metabolism of L-dopa. After E. faecalis converts the drug into dopamine, a second organism converts dopamine into another compound, meta-tyramine.
To find the second organism, Maini Rekdal experimented with a fecal sample. He subjected its diverse microbial community to a Darwinian game, feeding dopamine to hordes of microbes to see which prospered.
Eggerthella lenta won. These bacteria eat dopamine and make meta-tyramine as a byproduct, a challenging reaction even for chemists. “There’s no way to do it on the bench top,” Maini Rekdal said, “and previously no enzymes were known that did this exact reaction.”
Knowing the microbial meals end with meta-tyramine, Maini Rekdal decided to test whether he could predict how a fecal sample’s bacteria would interact with L-dopa.
He could. Based on meta-tyramine levels, Maini Rekdal determined how much of the drug a sample would consume. With this in mind, doctors could individualize Parkinson’s treatment based on a patient’s specific microbes and their byproducts.
Meta-tyramine may also contribute to L-dopa’s noxious side effects, but until now, there was no reason to investigate the compound’s role.
“All of this suggests that gut microbes may contribute to the dramatic variability that is observed in side effects and efficacy between different patients taking L-dopa,” said Balskus. Her work could help decrease this variability and allow the drug to work as intended, without microbial interference.








